
The surroundings will compensate accordingly to keep the energy of universe constant and increase the entropy. The energy (in the form of enthalpy) and entropy can increase or decrease in a system. Note that these two statements are talking about the system, rather than the universe. Based on these two laws, we can determine that statement I and statement II are false. 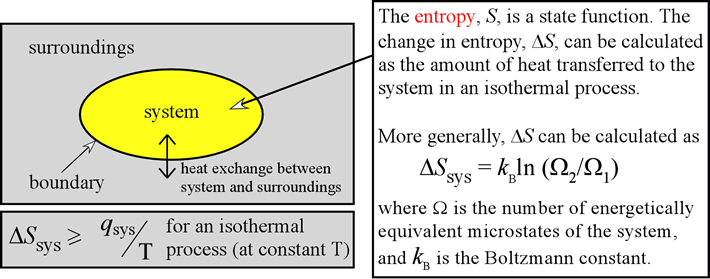
This suggests that the universe is always going towards a more disordered state. The second law of thermodynamics states that the entropy, or the amount of disorder in the universe, is always increasing. The energy lost by a system is gained by surroundings and vice versa however, the total energy of the universe is always constant. 
The first law of thermodynamics states that the energy of the universe is always constant, which implies that energy cannot be created or destroyed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
